News
Updates And Announcements

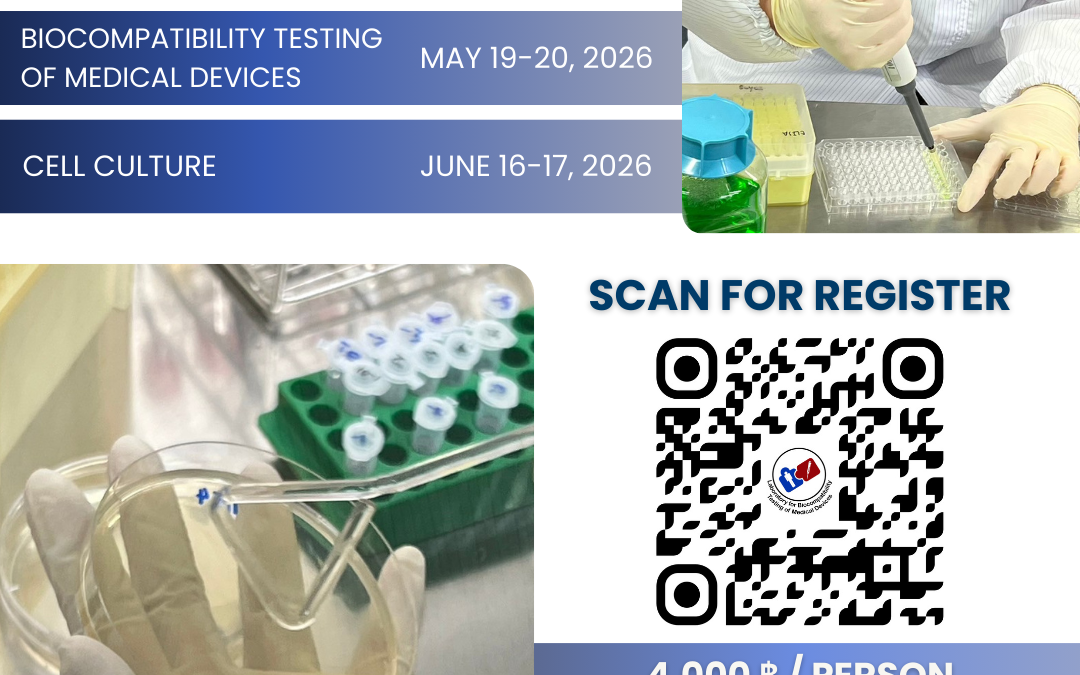
The 14th “Biocompatibility Testing of Medical Devices” Workshop
On May 19–20, 2026, the Department of Biomedical Engineering, Faculty of Engineering, Mahidol University, in collaboration with the ISO 17025-certified Biocompatibility Testing Laboratory for Medical Devices, organized a hands-on workshop on medical equipment and devices. The objective was to enhance the capabilities and workforce in the medical device industry sector. The laboratory conducted training sessions focused on Upskilling, Reskilling, and New skilling under the topic “Biocompatibility Testing of Medical Devices” for participants from both the public and private sectors.
Special Lecture on Comparative Medical Device Regulatory Frameworks in the U.S. and Asia
On March 17–18, 2026, at True Space Mahidol, Salaya, Nakhon Pathom, Thailand, the ISO 17025 Medical Device Biocompatibility Testing Laboratory, led by Assoc. Prof. Dr. Norased Nasongkla, Director of the Medical Device Biocompatibility Testing Laboratory (ISO 17025) and Director of the Drug Delivery System Laboratory for Medical Devices (ISO 13485), organized a Special Lecture on Comparative Medical Device Regulatory Frameworks in the U.S. and Asia.
Invitation to Participate in the 2025 Hands-on Training Workshops (Apr–Jun 2025)
Laboratory for Biocompatibility Testing of Medical Devices will organize hands-on training workshops on the following topics for Round 2 of 2026: (NOTE: Workshop is in Thai) Microbial Culture Topics: Basic Microbial Culture Microbial Purification Microbial...
The 16th Cell Culture Workshop
On June 10-11, 2025, the Department of Biomedical Engineering, Faculty of Engineering, Mahidol University, in collaboration with the ISO 17025-certified Biocompatibility Testing Laboratory for Medical Devices, organized a hands-on workshop on medical equipment and devices. The objective was to enhance the capabilities and workforce in the medical device industry sector. The laboratory conducted training sessions focused on Upskilling, Reskilling, and Newskilling under the topic “Cell Culture” for participants from both the public and private sectors.
Latest News
The 14th “Biocompatibility Testing of Medical Devices” Workshop
On May 19–20, 2026, the Department of Biomedical Engineering, Faculty of Engineering, Mahidol University, in collaboration with the ISO 17025-certified Biocompatibility Testing Laboratory for Medical Devices, organized a hands-on workshop on medical equipment and devices. The objective was to enhance the capabilities and workforce in the medical device industry sector. The laboratory conducted training sessions focused on Upskilling, Reskilling, and New skilling under the topic "Biocompatibility Testing of Medical Devices" for participants from both the public and private sectors.
Special Lecture on Comparative Medical Device Regulatory Frameworks in the U.S. and Asia
On March 17–18, 2026, at True Space Mahidol, Salaya, Nakhon Pathom, Thailand, the ISO 17025 Medical Device Biocompatibility Testing Laboratory, led by Assoc. Prof. Dr. Norased Nasongkla, Director of the Medical Device Biocompatibility Testing Laboratory (ISO 17025) and Director of the Drug Delivery System Laboratory for Medical Devices (ISO 13485), organized a Special Lecture on Comparative Medical Device Regulatory Frameworks in the U.S. and Asia.
Invitation to Participate in the 2025 Hands-on Training Workshops (Apr–Jun 2025)
Laboratory for Biocompatibility Testing of Medical Devices will organize hands-on training workshops on the following topics for Round 2 of 2026: (NOTE: Workshop is in Thai) Microbial Culture Topics: Basic Microbial Culture Microbial Purification Microbial Determination 1) Date: 28–29 April 2026 Biocompatibility Testing of Medical Devices Topics: Cytotoxicity Hemolysis Irritation and Sensitization 2) Date: 19–20 […]
The 16th Cell Culture Workshop
On June 10-11, 2025, the Department of Biomedical Engineering, Faculty of Engineering, Mahidol University, in collaboration with the ISO 17025-certified Biocompatibility Testing Laboratory for Medical Devices, organized a hands-on workshop on medical equipment and devices. The objective was to enhance the capabilities and workforce in the medical device industry sector. The laboratory conducted training sessions focused on Upskilling, Reskilling, and Newskilling under the topic "Cell Culture" for participants from both the public and private sectors.
Laboratory visitation from Thailand Advanced Institute of Science and Technology and the Tokyo Institute of Technology (TAIST–Science Tokyo)
On 30 January 2026, the Laboratory for Biocompatibility Testing of Medical Devices, led by Associate Professor Dr. Norased Nasongkla, welcomed a delegation from TAIST–Science Tokyo, with the visit serving to strengthen international academic and research collaboration.
Laboratory visitation for Dr. Donald Balance and Sophia Wong from the University of Glasgow
On January 29, 2026, the Laboratory for Biocompatibility Testing of Medical Devices welcomed Dr. Donald Balance and Sophia Wong from the University of Glasgow for a visit to the Laboratory for Biocompatibility Testing of Medical Devices, Department of Biomedical Engineering, Faculty of Engineering, Mahidol University.
Laboratory visitation for speakers Chiranya Prachaseri, CEO of Cryoviva (Thailand) Ltd.
On 11 November 2025, the Laboratory for Biocompatibility Testing of Medical Devices, led by Assoc. Prof. Dr. Norased Nasongkla, welcomed special guest speaker Chiranya Prachaseri, CEO of Cryoviva (Thailand) Ltd. She delivered a lecture on "Wellness: Business Perspectives Every Healthcare Professional Should Know" as part of the Thai Biomedical Engineering Special Seminar Series, and was also given a tour of the Laboratory for Biocompatibility Testing of Medical Devices, Department of Biomedical Engineering, Faculty of Engineering, Mahidol University.
Laboratory Visitation from Prof. M.L. Chagriya Kitiyakara Faculty of Medicine Ramathibodi Hospital, Mahidol University
August 7, 2025, the Laboratory for Biocompatibility Testing of Medical Devices, led by Associate Professor Dr. Norased Nasongkhla, welcomed visitors from the Faculty of Medicine Ramathibodi Hospital, Mahidol University, led by Prof. M.L. Chagriya Kitiyakara. The visit involved a tour of the Laboratory for Biocompatibility Testing of Medical Devices under the Department of Biomedical Engineering, Faculty of Engineering, Mahidol University
Participation in the exhibition at the event connecting research and services of Mahidol University to create business matching opportunities with the industrial sector
On June 30, 2025, the ISO 17025-certified Biocompatibility Testing Laboratory for Medical Devices participated in the exhibition at the event connecting research and services of Mahidol University to create business matching opportunities with the industrial sector. The event was organized by the Federation of Thai Industries (FTI), Industrial Group, in collaboration with the Institute for Technology and Innovation Management (iNT), Mahidol University, at the Queen Sirikit National Convention Center, Bangkok
Participation in the Mahidol University-Industry Partnerships Forum – Advanced Materials Technology and Innovation at Satharanat Mongkol Building, Faculty of Science, Phayathai, Mahidol University.
On June 11–13, 2025, the ISO 17025-certified Medical Device Biocompatibility Testing Laboratory participated in the 33rd Annual Medical Science Conference under the theme “Transforming Healthcare Through Modern Medical Sciences”, organized by the Department of Medical Sciences at IMPACT Forum, Muang Thong Thani, Nonthaburi Province.
Gallery
Our Past Work