Laboratory for Biocompatibility Testing of Medical Devices
World-Class Biocompatibility Testing Laboratory
for Medical Device Industry
ISO/IEC 17025:2017 Certified
World-class standards for the competency of the laboratory, a qualification all professional laboratories require.
Timely and Responsive
In matters of business and saving patients, time is of the essence. Our Fast Track service offers results within 14 days.
Knowledge Builds Capability
Clear, concise and complete: our training services adheres to this principles to raise the capabilities of our attendees.
A Global Standard
For World-Class Service
The Laboratory for Biocompatibility Testing of Medical Devices is a certified ISO/IEC 17025:2017 laboratory that specializes in tests such as for biocompatibility and cytotoxicity, for medical devices such as gauze.
The laboratory operates under the Faculty of Engineering, Mahidol University, and is certified under global standards.
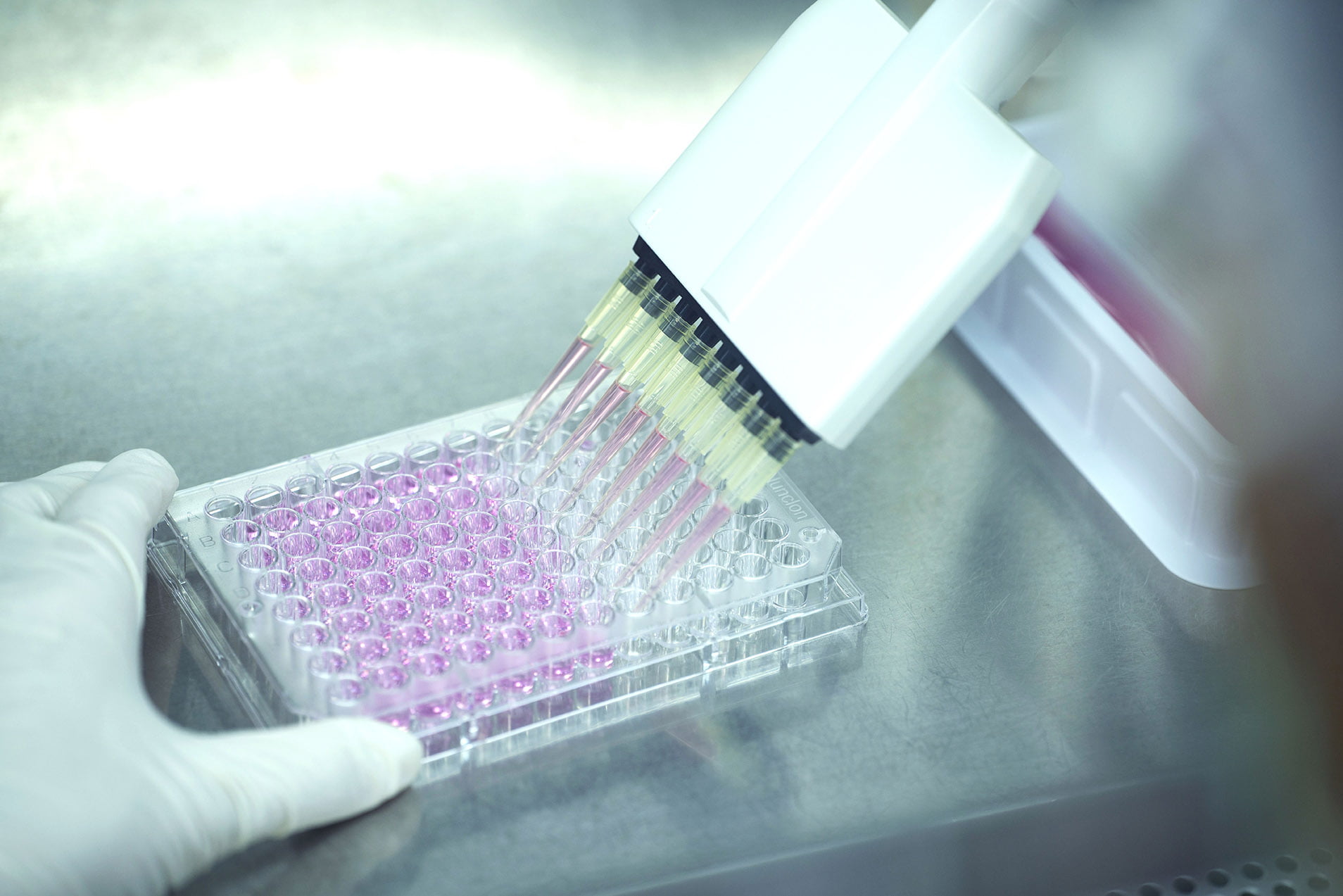
Testing Services for Medical Devices
Our services include cytotoxicity testing, hemolysis testing, microbiology testing, antibacterial susceptibility testing and composition analysis for medical devices.

Training Services for Industry Personnel
Our training modules for industry personnel and interested parties include topics such as working with cell cultures, biocompatibility testing of medical devices, and microbiology.
Our Latest News
Updates & Announcements
The Laboratory for Biocompatibility Testing of Medical Devices would like to present the results of our services, of which our customers were most satisfied with.
Special Lecture on Comparative Medical Device Regulatory Frameworks in the U.S. and Asia
On March 17–18, 2026, at True Space Mahidol, Salaya, Nakhon Pathom, Thailand, the ISO 17025 Medical Device Biocompatibility Testing Laboratory, led by Assoc. Prof. Dr. Norased Nasongkla, Director of the Medical Device Biocompatibility Testing Laboratory (ISO 17025) and Director of the Drug Delivery System Laboratory for Medical Devices (ISO 13485), organized a Special Lecture on Comparative Medical Device Regulatory Frameworks in the U.S. and Asia.
Invitation to Participate in the 2025 Hands-on Training Workshops (Apr–Jun 2025)
Laboratory for Biocompatibility Testing of Medical Devices will organize hands-on training workshops on the following topics for Round 2 of 2026: (NOTE: Workshop is in Thai) Microbial Culture Topics: Basic Microbial Culture Microbial Purification Microbial...

